Pheburane® Coated Oral Pellets:
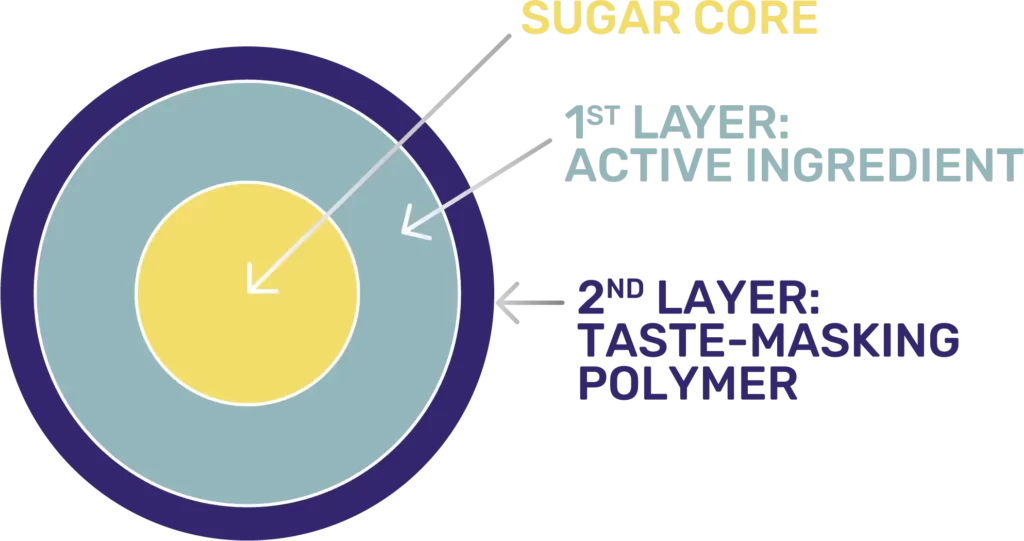
An innovative formulation of sodium phenylbutyrate (NaPB) with a special coating designed to mask the awful taste of NaPB
The FDA-approved safe, effective, and palatable adjunctive therapy to the standard of care, which includes dietary management, for the treatment of certain Urea Cycle Disorders (UCDs)
- An innovative alternative to other UCD treatments – no tablets, no syringes, & no mixing required.
- Measured with a reusable calibrated dosing spoon.
- Offered at a price that is similar to the other sodium phenylbutyrate (NaPB) products and significantly less expensive than the most commonly prescribed chronic UCD treatment.
- With Pheburane®, UCD patients found the ease of administration & acceptability much higher when compared to non-taste-masked NAPB products in a Cohort Temporary Utilization Authorization (ATU) Survey of Patients2. Please see Clinical Experience for additional study information.
Episodes of acute hyperammonemia may occur in patients while on Pheburane®. Pheburane® is not indicated for the treatment of acute hyperammonia, which can be a life-threatening medical emergency that requires rapid acting interventions to reduce plasma ammonia levels. Pheburane® treatment should be supervised by a healthcare provider experienced in the treatment of UCDs.
PHEBURANE® PELLETS: A polymer coating designed to mask the awful taste of NaPB
P=Palatable
P=Pellets
Impact of Urea Cycle Disorders4,5
Urea Cycle Disorders (UCDs) are a group of rare inherited metabolic diseases characterized by deficiencies of one of the enzymes and transporters involved in the urea cycle. The urea cycle is necessary for the removal of nitrogen produced from the breakdown of protein. UCDs are characterized by hyperammonemia and life-threatening hyperammonemic crises.5
In UCDs, if patients fail to comply with their NaPB treatment, it may result in symptoms of poor control of plasma ammonia levels that may lead to potentially irreversible and life-threatening situations.4
PHEBURANE®P=Proven
Study Title: Results from a Nationwide Cohort Temporary Utilization Authorization (ATU) Survey of Patients in France Treated with Pheburane® (sodium phenylbutyrate) Taste-Masked Granules2†
NO hyperammonemic episodes were experienced in the Pheburane® study treatment group (n=10).
Pheburane® treatment led to significantly lower plasma ammonia (NH3) and glutamine (Gln) values compared to non-taste-masked NaPB treatments.
- Based on temporary utilization authorization (ATU) protocol in France to analyze safety and efficacy in the treated cohort of patients with UCD.
- The FDA-approved term pellets is used to describe Pheburane® in the United States. The term granules is used to describe Pheburane® in other countries.
The most common adverse reactions associated with Pheburane® (incidence ≥ 3%) are menstrual dysfunction, decreased appetite, body odor and bad taste or taste aversion.
How to Take Pheburane®1,5
Dietary restriction of protein combined with an ammonia-scavenging treatment is one of the main therapeutic modalities used in the chronic treatment of UCDs. NaPB has been the standard treatment of UCDs for over 25 years.
Pheburane® is an innovative taste-masked formulation of NaPB, indicated as adjunctive therapy to the standard of care which includes dietary management for the treatment of certain UCDs
2 Ways Pheburane® can be taken with food consumption (meal or snack):
- swallowed with a beverage – e.g., water, fruit juices, protein-free infant formulas
- sprinkled on soft food – apple sauce or carrot puree* – immediately before eating
* Administration of Pheburane® oral pellets with other foods has not been studied and is not recommended. Additionally, administration with soft food is only recommended in patients old enough to consume soft foods.
Patients should swallow pellets immediately (within 10 seconds) to minimize dissolution of coating. Please see Instructions for Use.

10 Second Window of Opportunity
The in vitro test profile of Pheburane® coated pellets indicates that the NaPB was only released after approximately 10 seconds followed by a slow release over several minutes. Therefore, patients must take as directed and optimally within 10 seconds to avoid any bad taste.4
Pheburane®P=PRICE
Medunik USA offers Pheburane® through its UNIK Support Program – designed to support each unique patient. Unik Support (pronounced yoo·neek) has specialized services for Prescribers and Patients, including a Savings Program.
Medunik USA provides Pheburane® and all its added benefits at a cost that is similar to the other NaPB products and significantly less expensive than the most commonly prescribed chronic UCD treatment.
Important Safety Information
Pheburane® is indicated as adjunctive therapy to standard of care, which includes dietary management, for the chronic management of adult and pediatric patients with urea cycle disorders (UCDs), involving deficiencies of carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC) or argininosuccinic acid synthetase (AS).
Limitations of Use
Episodes of acute hyperammonemia may occur in patients while on Pheburane®. Pheburane® is not indicated for the treatment of acute hyperammonemia, which can be a life-threatening medical emergency that requires rapid acting interventions to reduce plasma ammonia levels. Read more
Important Safety Information
Pheburane® is indicated as adjunctive therapy to standard of care, which includes dietary management, for the chronic management of adult and pediatric patients with urea cycle disorders (UCDs).
Limitations of Use
Pheburane® is not indicated for the treatment of acute hyperammonemia. Read more
Important Safety Information
INDICATION
Pheburane® is indicated as adjunctive therapy to standard of care, which includes dietary management, for the chronic management of adult and pediatric patients with urea cycle disorders (UCDs), involving deficiencies of carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC) or argininosuccinic acid synthetase (AS).
Limitations of Use
Episodes of acute hyperammonemia may occur in patients while on Pheburane®. Pheburane® is not indicated for the treatment of acute hyperammonemia, which can be a life-threatening medical emergency that requires rapid acting interventions to reduce plasma ammonia levels.
WARNINGS AND PRECAUTIONS
Neurotoxicity of Phenylacetate
Increased exposure to phenylacetate, the major metabolite of Pheburane®, may be associated with neurotoxicity in patients with UCDs. If symptoms of vomiting, nausea, headache, somnolence or confusion are present in the absence of high ammonia levels, consider reducing the dose of Pheburane®.
Hypokalemia
Renal excretion of phenylacetylglutamine may induce urinary loss of potassium. Monitor serum potassium during therapy, and initiate appropriate treatment when necessary.
Conditions Associated with Edema
In order to decide if administration of Pheburane® is appropriate in patients with diseases that involve edema, calculate the total amount of sodium patients will be exposed to, based on their weight or body surface area. If a patient develops new-onset edema or worsening edema while on treatment, discontinue administration of Pheburane® and initiate appropriate therapy.
Diabetes Mellitus, Hereditary Fructose Intolerance, Glucose-Galactose Malabsorption or Sucrase-Isomaltase Insufficiency
Avoid use of Pheburane® in patients with rare hereditary problems of fructose intolerance, glucose-galactose malabsorption or sucrose-isomaltase insufficiency.
ADVERSE REACTIONS
The most common adverse reactions associated with the use of Pheburane® (incidence > 3%) are menstrual dysfunction, decreased appetite, body odor and bad taste or taste aversion.
DRUG INTERACTIONS
- Valproic acid, Haloperidol, or Corticosteroids: May increase plasma ammonia levels. Monitor ammonia levels closely.
- Probenicid: May inhibit renal excretion of metabolites of Pheburane® including phenylacetate and phenylacetylglutamine. Monitor patients for potential neurotoxicity.
OVERDOSAGE
Overdoses of Pheburane® exceeding ten-fold the maximum recommended dosage may produce emesis, CNS depression, metabolic acidosis with or without respiratory alkalosis, hypernatremia, hypokalemia, and hypophosphatemia. Symptoms of overdose overlap with those of acute hyperammonemia. If overdose occurs, discontinue Pheburane®, monitor plasma phenylacetate and ammonia levels closely, and institute appropriate emergency management.
To report suspected adverse reactions, contact Medunik USA at 1-844-884-5520 or [email protected].
Please read the Full Prescribing Information.References:
- Pheburane® (sodium phenylbutyrate) oral pellets [Prescribing Information]. Medunik USA, Inc.
- Kibleur Y, Dobbelaere D, Barth M, Brassier A, Guffon N. Results from a Nationwide Cohort Temporary Utilization Authorization (ATU) survey of patients in france treated with Pheburane® (Sodium Phenylbutyrate) taste-masked granules. Paediatr Drugs. 2014.
- Kibleur Y, Guffon N. Long-Term Follow-Up on a Cohort Temporary Utilization Authorization (ATU) Survey of Patients Treated with Pheburane (Sodium Phenylbutyrate) Taste-Masked Granules. Paediatr Drugs. 2016 Apr;18(2):139-44. doi: 10.1007/s40272-015-0159-8. PMID: 26747635.
- Guffon N, Kibleur Y, Copalu W, Tissen C, Breitkreutz J. Developing a new formulation of sodium phenylbutyrate. Arch Dis Child. 2012 Dec;97(12):1081-5. doi: 10.1136/archdischild-2012-302398. Epub 2012 Aug 31. PMID: 22941860.
- Peña-Quintana L, Llarena M, Reyes-Suárez D, Aldámiz-Echevarria L. Profile of sodium phenylbutyrate granules for the treatment of urea-cycle disorders: patient perspectives. Patient Prefer Adherence. 2017 Sep 6;11:1489-1496. doi: 10.2147/PPA.S136754. PMID: 28919721; PMCID: PMC5593420.



